Common plastic wastes will be fragmented into microplastics over time, which cannot be biologically assimilated, thus causing persistent pollution and damage to humans and animals. By contrast, bacterial polyhydroxyalkanoates (PHA), a class of diverse and natural intracellular biopolyesters accumulated as carbon and energy storage materials, have been shown to be biodegradable and biocompatible. Thus, PHA are increasingly attractive as eco-friendly biodegradable plastics for replacing petrochemical plastics.
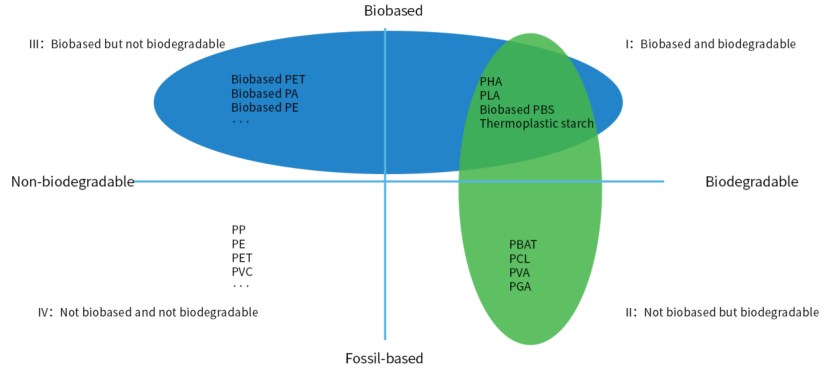
Fig. 1. Types of bioplastics with examples[1].
The advantages of PHA include excellent and controllable biodegradability, inherent biocompatibility, diverse mechanical properties and thermo-processability[2,3]. Benefiting from these features, PHA can be developed for medical microspheres, medical slow-release carriers, human implantation materials, antibacterial fibers and feeds.
Several studies have provided evidence that PHA is suitable as implanted scaffolds for bone tissue engineering to promote cellular osteogenesis. A study entitled "Enhanced bone regeneration via PHA scaffolds coated with polydopamine-captured BMP2" combined the advanced 3D printing technique with a simple functionalized process to create functionalized 3D-printed PHA scaffolds as a versatile platform for bone tissue regeneration. The P34HB-related biodegradable product 3-hydroxybutyrate(3HB) exhibited an inherent osteoinductive property, which may help P34HB scaffolds to promote bone regeneration to some extent[4,5].
In another study, PHA was used to prepare highly open porous microspheres(OPMs) of 300–360 µm in diameter, combining the advantages of microspheres and scaffolds to serve as injectable carriers harboring proliferating stem cells[6]. In addition to the convenient injection to a defected tissue, PHA OPMs protect cells against stresses during injection, allowing more living cells to proliferate and migrate to damaged tissues.
The studies above showed the advantage of PHA's biocompatibility and function of tissue regeneration. While other studies focus on d-β-hydroxybutyrate (d-3HB), which is a monomer of microbial poly-d-β-hydroxybutyrate (PHB). It is also a natural ketone body produced during carbohydrate deprivation to provide energy to the body cells, heart, and brain.

Fig. 2. Medical use of 3-hydrocybutyric acid (3HB) [7].
In recent years, increasing evidence demonstrates that d-3HB can induce pleiotropic effects on the human body, which are highly beneficial for improving physical and metabolic health. The benefits of endogenous d-3HB include improving performance in ultra-endurance events[8] , enhancing body composition[9], reducing levels of bad LDL cholesterol[10], altering metabolism-related hormones[11], suppressing appetite[12] and maintaining intestinal health status[13].
Specifically, lifestyle alteration is a key strategy used to tackle obesity and long-term KDs (ketogenic diet) or very low-energy diets have already been explored as a promising treatment strategy. d-3HB was shown to be able to improve the blood lipid profile in obese adults by a reduction in low density lipoprotein (LDL) cholesterol, increase in high density lipoprotein (HDL) cholesterol, smaller adipocyte cell volume, and inhibition of lipolysis via a G-protein-coupled receptor (GPCR) which reduced subsequent release of serum lipolytic products[14]. At the cellular level, d-3HB markedly increased mitochondrial uncoupling in brown adipose tissue (BAT) which increased mitochondrial respiration and thermogenesis, thereby resulting in increased resting energy expenditure (REE) in the obese[15,16].
A number of studies have investigated the pathways by which d-3HB acts to ameliorate Alzheimer’s and Parkinson’s Disease in rodent models[17,18]. In fact, in one clinical report of a patient with sporadic ALZ following treatment with KME, the patient demonstrated remarkable improvements in mood, self-caring ability, cognitive, and physical performance[19].
d-3HB also shows importance in protecting animals against a variety of bacterial diseases for growth. Clear evidence indicating that PHB conferred protection to Artemia host against V. campbellii by a mechanism of inducing heat shock protein (Hsp) 70[20]. PHB also has been studied as a feed additive for large yellow croakers and weaned piglets. As fish feed additive for growing large yellow croakers, PHB could increase fish weights 10-25%[21].

Fig. 3. PHB as safe food additive [22].
In recent years, the use of exogenous ketone supplements, which induce rapid ketosis without the need to adhere to the KD, has increased in popularity. Ingestion of d-3HB could effectively optimize the body composition of athletes, leading to enhanced exercise performance. However, ingestion of ketone salts and esters was associated with unwanted GI side effects or requires further degradation before releasing d-3HB for absorption. To our knowledge, there has been no direct investigation based on supplementing the free acid form of d-3HB as a beverage drink to humans or animals yet due to difficulties in obtaining the free acid form in large amounts.
To date, the majority of patented methods related to the production of exogenous d-3HB were through chemical synthesis [23]. While recently, PHB could be efficiently produced through bacterial synthesis using a new strain of bacteria known as Halomonas TD01[24]. Hence, the recently patented technology which utilizes a biosynthetic process to produce d-3HB acid on a kilogram scale is considered a first-time success within this field.
In conclusion, PHA and d-3HB have great potential in medical application with their inherent biocompatibility. We hope that the availability of PHA and d-3HB acid on a large scale may attract and aid scientists and researchers worldwide in developing novel treatments which will benefit the overall metabolic health of humans and potentially extend human life expectancy.
Reference:
1. SUSlick, K. S. Kirk-Othmer encyclopedia of chemical technology, 1998.
2. Z. Luo, Y. L. Wu, Z. B. Li and X. J. Loh, Recent Progress in Polyhydroxyalkanoates-Based Copolymers for Biomedical Applications, Biotechnol. J., 2019, 14, 1900283.
3. J. Y. Zhang, E. I. Shishatskaya, T. G. Volova, L. F. da Silva and G. Q. Chen, Polyhydroxyalkanoates (PHA) for therapeutic applications, Mater. Sci. Eng., C, 2018, 86, 144–150.
4. Y. Zhao, B. Zou, Z. Y. Shi, Q. Wu and G. Q. Chen, The effect of 3-hydroxybutyrate on the in vitro differentiation of murine osteoblast MCM-E1 and in vivo bone formation in ovariectomized rats, Biomaterials, 2007, 28, 3063–3073.
5. Q. Cao, J. Y. Zhang, H. T. Liu, Q. Wu, J. C. Chen and G. Q. Chen, The mechanism of anti-osteoporosis effffects of 3-hydroxybutyrate and derivatives under simulated microgravity, Biomaterials, 2014, 35, 8273–8283.
6. Dai-Xu Wei, Jin-Wei Dao, and Guo-Qiang Chen, A Micro-Ark for Cells: Highly Open Porous Polyhydroxyalkanoate Microspheres as Injectable Scaffolds for Tissue Regeneration, 2018, 30, 1802273.
7. Mierziak, Justyna et al. “3-Hydroxybutyrate as a Metabolite and a Signal Molecule Regulating Processes of Living Organisms.” Biomolecules, 2021, vol. 11,3 402.9.
8. Chang CK, Borer K, Lin PJ. Low-carbohydrate high-fat diet: can it help exercise performance? J Hum Kinetics, 2017, 56: 81-92.
9. Vargas S, Romance R, Petro JL, et al. Efficacy of ketogenic diet on body composition during resistance training in trained men: a randomized controlled trial. J Int Soc Sports Nutr, 2018, 15: 31.
10. Choi HR, Kim J, Lim H, et al. Two-week exclusive supplementation of modified ketogenic nutrition drink reserves lean body mass and improves blood lipid profile in obese adults: a randomized clinical trial. Nutrients, 2018, 10: 1895.
11. Sumithran P, Prendergast LA, Delbridge E, et al. Ketosis and appetite-mediating nutrients and hormones after weight loss. Eur J Clin Nutr, 2013, 67(7): 759-764.
12. Mohorko N, Černelič-Bizjak M, Poklar-Vatovec T, et al. Weight loss, improved physical performance, cognitive function, eating behavior, and metabolic profile in a 12-week ketogenic diet in obese adults. Nutr Res, 2019, 62: 64-77.
13. Cheng CW, Biton M, Haber AL, et al. Ketone body signaling mediates intestinal stem cell homeostasis and adaptation to diet. Cell, 2019, 178(5): 1115-1131.
14. Caminhotto RDO, Komino ACM, de Fatima SF, Andreotti S, Sertié RAL, Boltes Reis G, Lima FB (2017) Oral β-hydroxybutyrate increases ketonemia, decreases visceral adipocyte volume and improves serum lipid profle in Wistar rats. Nutr Metab (lond) 14:31–40.
15. Deemer SE, Davis RAH, Roberts BM, Smith DL, Koutnik AP, Poff AM, D’Agostino DP, Plaisance EP (2020) Exogenous dietary ketone ester decreases body weight and adiposity in mice housed at thermoneutrality. Obesity 28:1447–1455.
16. Walton CM, Jacobsen SM, Dallon BW, Saito ER, Bennett SLH, Davidson LE, Thomson DM, Hyldahl RD, Bikman BT (2020) Ketones elicit distinct alterations in adipose mitochondrial bioenergetics. Int J Mol Sci 21:1–14.
17. Kashiwaya Y, Takeshima T, Mori N, Nakashima K, Clarke K, Veech RL (2000) D-beta -Hydroxybutyrate protects neurons in models of Alzheimer’s and Parkinson’s disease. Proc Natl Acad Sci 97:5440–5444.
18. Wu Y, Gong Y, Luan Y, Li Y, Liu J, Yue Z, Yuan B, Sun J, Xie C, Li L, Zhen J, Jin X, Zheng Y, Wang X, Xie L, Wang W (2020b) BHBA treatment improves cognitive function by targeting pleiotropic mechanisms in transgenic mouse model of Alzheimer’s disease. FASEB J 34:1412–1429.
19. Newport MT, VanItallie TB, Kashiwaya Y, King MT, Veech RL (2015) A new way to produce hyperketonemia: use of ketone ester in a case of Alzheimer’s disease. Alzheimers Dement 11:99–103.
20. Yafei Duan, Yue Zhang, Hongbiao Dong, Xiaoting Zheng, Yun Wang, Hua Li, Qingsong Liu, Jiasong Zhang, Effect of dietary poly-β-hydroxybutyrate (PHB) on growth performance, intestinal health status and body composition of Pacific white shrimp Litopenaeus vannamei (Boone, 1931), Fish & Shellfifish Immunology 60 (2017) 520-528.
21. Xuan Wang, Xiao-Ran Jiang, Fuqing Wu, Yiming Ma, Xuemei Che, Xiyue Chen, Ping Liu, Wenbing Zhang, Xi Ma, Guo-Qiang Chen, Microbial Poly-3-Hydroxybutyrate (PHB) as a Feed Additive for Fishes and Piglets. Biotechnol J. 2019 Dec;14(12):e1900132.
22. Wang, X., Jiang, X. R., Wu, F., Ma, Y., Che, X., Chen, X., ... & Chen, G. Q. Microbial poly‐3‐hydroxybutyrate (PHB) as a feed additive for fishes and piglets. Biotechnology Journal, 2019, 14(12), 1900132.
23. Haas T, Hecker A, Potter M, Bulter T (2018) A method of synthesizing 3-hydroxybutyric acid. [Patent No.WO2017/016902]、
24. Tan D, Xue Y-S, Aibaidula G, Chen G-Q (2011) Unsterile and continuous production of polyhydroxybutyrate by Halomonas TD01. Bioresour Technol 102:8130–8136.